Skin’s innate immunity is the initial activator of immune response mechanisms, influencing the development of adaptive immunity. Some contact allergens are detected by Toll-like receptors (TLRs) and inflammasome NLR3. Keratinocytes participate in innate immunity and, in addition to functioning as an anatomical barrier, secrete cytokines, such as TNF, IL-1β, and IL-18, contributing to the development of Allergic Contact Dermatitis. Dendritic cells recognize and process antigenic peptides into T cells. Neutrophils cause pro-inflammatory reactions, mast cells induce migration/maturation of skin DCs, the natural killer cells have natural cytotoxic capacity, the γδ T cells favor contact with hapten during the sensitization phase, and the innate lymphoid cells act in the early stages by secreting cytokines, as well as act in inflammation and tissue homeostasis. The antigen-specific inflammation is mediated by T cells, and each subtype of T cells (Th1/Tc1, Th2/Tc2, and Th17/Tc17) activates resident skin cells, thus contributing to inflammation. Skin’s regulatory T cells have a strong ability to inhibit the proliferation of hapten-specific T cells, acting at the end of the Allergic Contact Dermatitis response and in the control of systemic immune responses. In this review, we report how cutaneous innate immunity is the first line of defense and focus its role in the activation of the adaptive immune response, with effector response induction and its regulation.
Allergic Contact Dermatitis (ACD) is a generally eczematous, inflammatory skin disease, which begins at the location of contact with the allergen, after sensitization occurs, which is constituted by successive exposure to the allergen, provoking the activation of the immune system.1,2 The occurrence of cutaneous manifestations clinically represents the elicitation stage, followed by the immune resolution stage.2
Currently, it is known that, in ACD, the immune mechanisms involve both innate and adaptive immunity, and that, in the sensitization stage, the mere presence of the antigen is insufficient to generate an immune response, which requires the activation of the innate immune system.3
The contact allergens are low-weight molecules (less than 500 doltons), which needs to be linked, by covalent bonds, to a protein in the epidermis to generate a new antigenic determinant.3,4 By contrast, the sensitization to the metallic ions (nickel, chrome, cobalt, etc.) generate non-covalent bonds, which are capable of stimulating the immune response.4 The hapten induces sensitization due to its pro-inflammatory properties, with the capacity to activate the innate immunity of the skin and emit signals that induce the recruitment, migration, and maturation of Dendritic Cells (DCs).4
Two phases occur for the development of ACD: sensitization and elicitation.3
Sensitization stage (afferent stage or induction stage): this occurs after weeks or months of repeated exposure of the sensitizing agent to the skin.3 The compounds formed between the hapten and a protein are processed by the skin’s DCs, migrate to the Draining Lymph Nodes (dLNs), from which, in their paracortical regions, the antigen-specific T CD8+ and CD4+ lymphocytes originate, differentiating them from Th1 and Th17 effector cells and Cytotoxic T Lymphocytes (CTLs). This phase occurs between 10 and 15 days.3-5
Elicitation stage (efferent stage or effector stage): In this stage, the reexposure to the same hapten leads to the appearance of ACD through the recruitment of the Th1 and Th17 cells and CTLs to the skin, in which the CTLs induce the apoptosis of the keratinocytes. At the same time, there is the generation of the regulator T CD4+ cells (Tregs), capable of inhibiting ACD, mediating the tolerance in unallergic individuals.3-5 This stage can last from 24 to 72 hours, presenting clinical signs of inflammation.6,7 These appear as a cutaneous eruptive process, which can fall under many clinical modalities: erythematous-vesicular lesions or erythematous-vesicular-secreting lesions or erythematous-secreting-infiltrative-lichenified lesions, with pruritus representing a constant symptom of variable intensity.8
Resolution stage:evidence has shown that the Tregs control the expansion of T CD8+ cells in the lymph node and its activation in the skin.3
In this review, we report how the contact allergens promote inflammation through the activation of innate immunity, its cooperation amongst them, and with T cells to begin and guide early responses to contact allergens and the actions of Treg cells in the control of cutaneous inflammation (Chart 1).
Main abbreviations
| • Dendritic Cells (DCs) |
| • Dermal Dendritic Cells (dDCs) |
| • Plasmacytoid Dendritic Cells (pDCs) |
| • Langerhans Cells (LCs) |
| • Draining Lymph Nodes (dLNs) |
| • Cytotoxic T Lymphocytes (CTLs) |
| • Regulator T CD4+ cells (Tregs) |
| • Pathogen-Associated Molecular Patterns (PAMPs) |
| • Pattern Recognition Receptors (PRRs) |
| • Damage-Associated Molecular Patterns (DAMPs) |
| • Toll-like Receptors (TLRs) |
| • Antigen-Presenting Cells (APCs) |
| • Hyaluronic Acid (HA) |
| • Nickel (Ni2+) |
| • NLR3 NOD-Like Receptor (NLR) |
| • Adenosine Triphosphate (ATP) |
| • Natural Killer (NK) Cells |
| • T-Cell Receptors (TCRs) |
| • Innate Lymphoid Cells (ILCs) |
| • Thymic Stromal Lymphopoetin (TSLP) |
| • Granulocyte-Macrophage Colony-Stimulating Factor (GM-CSF) |
| • Regulatory T Cells-1 (Tr1) |
Cutaneous innate immunity constitutes the first line of defense as well as plays a key role in the activation of the adaptive immune response, which represents the second line of defense.9
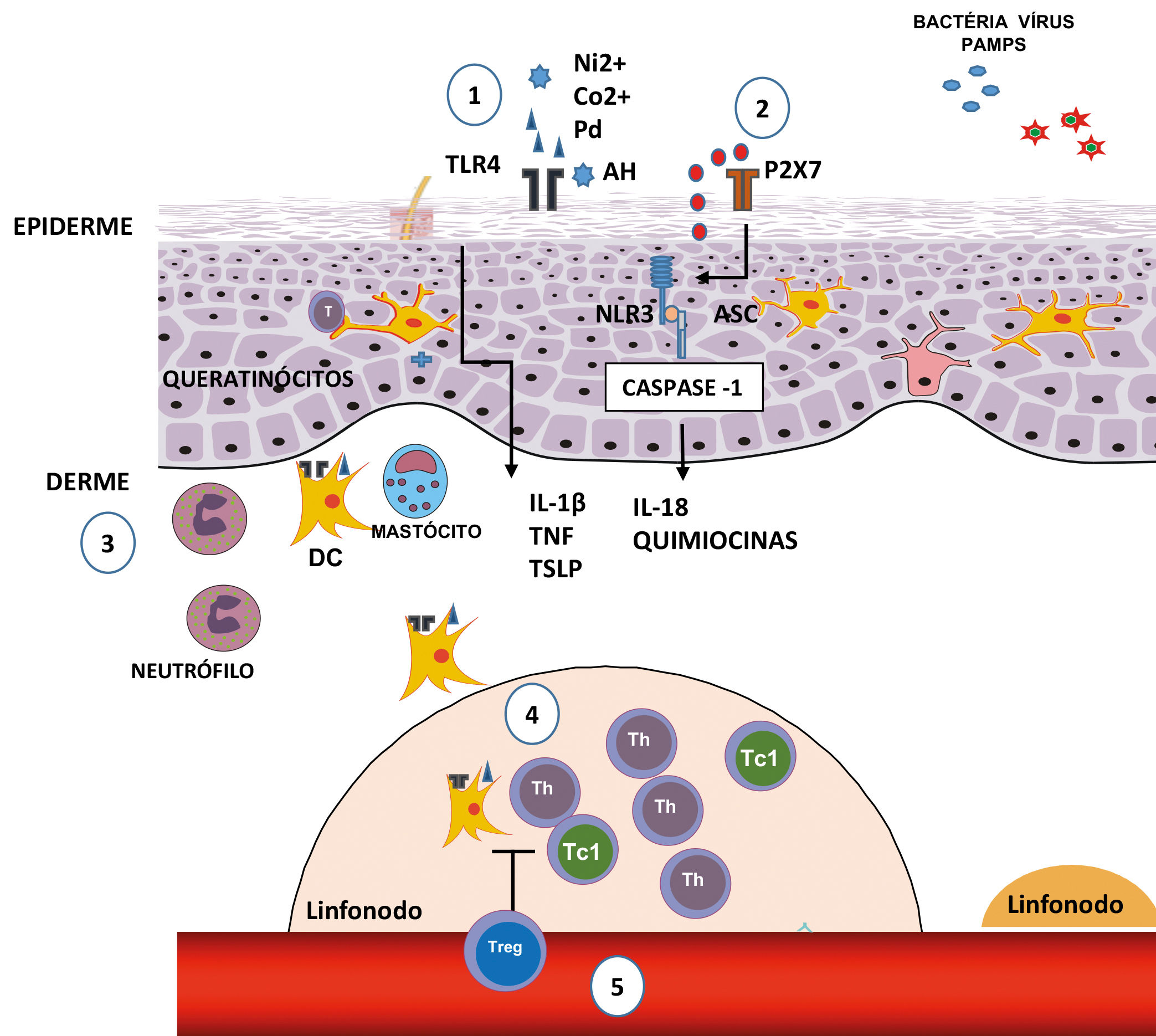
Innate immunity is characterized by its ability to recognize pathogens, such as viruses, bacteria, and fungi, that is, Pathogen-Associated Molecular Patterns (PAMPs), through a limited number of receptors, called Pattern Recognition Receptors (PRRs).10,11 These are expressed by various cell types, including macrophages, monocytes, DCs, neutrophils, keratinocytes, and epithelial cells, and allow for an early detection of pathogens in the location of infection (Figure 1).11
Initial stages of the sensitization in contact dermatites. 1) The nickel (Ni2+), cobalt (Co2+) and palladium (Pd) ions, or fragments of hyaluronic acid (HA) generated by some contact allergens, can directly trigger TLR4 and the NFκB pathway, culminating in the production of pro-inflammatory cytokines and chemokines. 2) The extracellular ATP acts as a danger signal (DAMP); upon binding with the P2X7 perigenetic receptor, it triggers the inflammasome through NLR3 and caspase-1, promoting the maturation of IL-1β and IL-18. 3) The production of cytokines and chemokines, by the keratinocytes, and the activation through TLR4 in DCs favors their maturation and migration, which is optimized with the presence of neutrophils and mast cells. 4) In dLNs, the DCs present the antigen for the induction of antigen-specific T effector cells, which can generate Th or T CD8+ cells which secrete IFN-γ (Tc1). 5) The regulation of Tregs in the sensitization stage, acting in the antigenic presentation and/or in the generation of effector T cells
The PRRs are able to recognize the Damage-Associated Molecular Patterns (DAMPs), which are molecules that can be released during cell death.3 DAMPs include a group of many proteins, nucleic acids, and glycosaminoglycans. DAMPs and PRRs play a key role in ACD.1,12
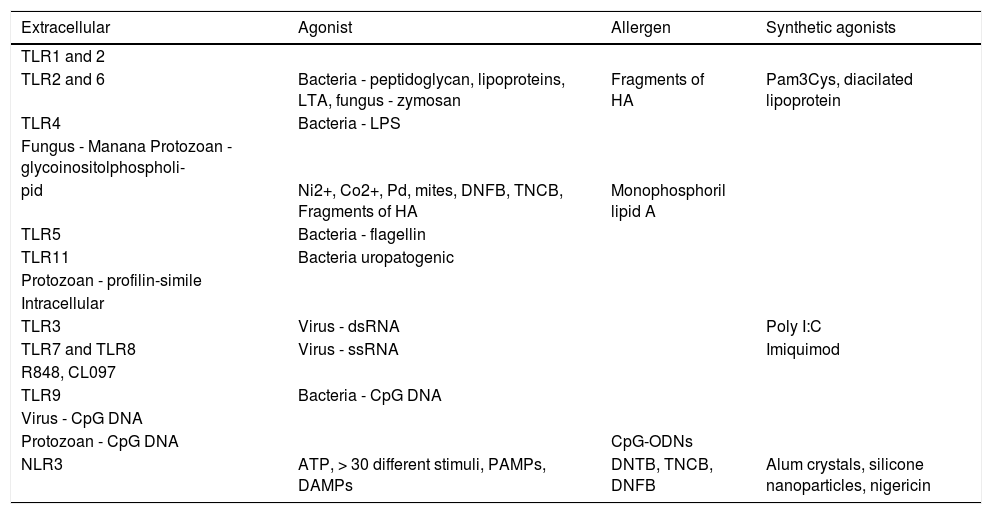
Toll-like Receptors (TLRs)TLRs are a family of receptors that recognize a wide variety of bonds, including lipids, lipoproteins, proteins, and nucleic acids.3 These receptors are expressive, especially in Antigen-Presenting Cells (APCs), epithelial, endothelial, and keratinocyte cells, working as a bond between innate and specific immunity.13,14 Have been identified 10 TLRs in humans and 12 in mice.15 The TLRs are expressed extracellularly as TLR 1, 2, 4, 5, 6, and 11, which mainly recognize lipids, lipoproteins, and proteins.16 By contrast, other TLRs are expressed intracellularly, in the endoplasmic reticulum, endosomes, lysosomes, and endolysosomes, as TLR 3, 7, 8, and 9, which recognize microbial nucleic acids.15,16
Each TLR recognizes a standard component, such as the TLR4, which recognizes the lipopolysaccharide (LPS), and its signaling induces the secretion of type 1 IFNs and other pro-inflammatory cytokines.11,13,16 By contrast, activation through other TLRs, such as the heterodimers of TLR1-TLR2 and TLR2-TLR6 or the homodimer of TLR5, leads to the secretion of pro-inflammatory cytokines, but without the production of IFNs.3
The heterodimers TLR1-TLR2 and TLR2-TLR6 recognize bacteria and micoplasmas. TLR3 acts in the prevention of viral infections, by recognizing the double ribbon of RNA (dsRNA).15 TLR7 and 8 recognize the double ribbon of viral RNA (ssRNA); TLR9 recognizes the components containing CpG in the DNA, and TLR11 recognizes uropathogenic bacteria (Chart 2).14
Toll-like receptor and inflammasome bonds
| Extracellular | Agonist | Allergen | Synthetic agonists |
|---|---|---|---|
| TLR1 and 2 | |||
| TLR2 and 6 | Bacteria - peptidoglycan, lipoproteins, LTA, fungus - zymosan | Fragments of HA | Pam3Cys, diacilated lipoprotein |
| TLR4 | Bacteria - LPS | ||
| Fungus - Manana Protozoan - glycoinositolphospholi- | |||
| pid | Ni2+, Co2+, Pd, mites, DNFB, TNCB, Fragments of HA | Monophosphoril lipid A | |
| TLR5 | Bacteria - flagellin | ||
| TLR11 | Bacteria uropatogenic | ||
| Protozoan - profilin-simile | |||
| Intracellular | |||
| TLR3 | Virus - dsRNA | Poly I:C | |
| TLR7 and TLR8 | Virus - ssRNA | Imiquimod | |
| R848, CL097 | |||
| TLR9 | Bacteria - CpG DNA | ||
| Virus - CpG DNA | |||
| Protozoan - CpG DNA | CpG-ODNs | ||
| NLR3 | ATP, > 30 different stimuli, PAMPs, DAMPs | DNTB, TNCB, DNFB | Alum crystals, silicone nanoparticles, nigericin |
PAMPs: Pathogen-Associated Molecular Patterns; DAMPs: Damage-Associated Molecular Patterns; Ni2+: nickel; Co2+: cobalt; Pd: palladium; HA: hyaluronic acid; LTA: lipoteichoic acid; LPS: lipopolysaccharide; dsRNA: double ribbon RNA; ssRNA: single ribbon RNA; CpG-ODNs: oligodeoxinucleotides containing CpG motives, household dust mites; DNTB: dinitrotiocianobenzene; TNCB: trinitrochlorobenzene; DNFB: dinitrofluorobenzene; NLR3: inflammasome; ATP: adenosine triphosphate.
There are two possible mechanisms of sensitization to contact allergens: one is the dependent IL-12, in which the isolated activation of TLR2 or TLR4 is enough to develop ACD, and an independent IL-12 mechanism, which requires the activation of both TLR2 and TLR4.3,17 TLR2 and TLR4 do not directly recognize the hapten, but these are able to recognize endogenous bonds that are present in the skin.3,12 The low-wieght molecules, derived from hyaluronic acid (HA), activate the DCs in vitro via TLR2 and TLR4, while the degradated HA acts as an endogenous activator of TLR2 and TLR4, signaling the ACD response.3
The nickel ion (Ni2+) is one of the most common causes of ACD.18 It directly activates TLR4 in such a way that the transgenic expression of TLR4 in an individual that lacks this results in an effective sensitization, indicating the crucial role of TLR4 in allergies to Ni2+.18,19 In addition to Ni2+, cobalt (Co2+) and palladium (Pd) ions can also directly activate TLR4, inducing its dimerization and inflammatory signaling through the Nuclear Factor-κB (NF-κB), culminating in the production of pro-inflammatory cytokines and chemokines.1,20
InflammassomeThe inflammasome is an intracelular protein complex, formed after the recognition of many inflammatory signals.3 The contact allergens activate the innate immune system through a complex mechanism involving TLRs, the NLR3 inflammasome (members of the NOD-Like Receptors (NLRs) family), and signs of endogenous damage, such as the extracellular Adenosine Triphosphate (ATP) and reactive oxygen species (ROS).2,17,19 The extracellular ATP can act as a danger signal (DAMP) upon bonding to the purinergic receptor P2X7, activating the inflammasome and promoting the maturation of IL-1β.21 The ATP, the main carrier of energy within the cell, and the P2X7 are essential for the activation of the NLR3 inflammasome by the contact allergen.22
The inflammasome leads to the activation of caspase-1, which cleaves pro-IL-1β and pro-IL-18, generating the active pro-inflammatory cytokines.11,16 The contact sensitizers, such as haptens, when applied to the skin, induce the processing and secretion of IL-1β and IL-18, dependent on inflammasomes, causing the ACD reaction.23 Thus, the exposure of the skin to hapten or high doses of UV radiation activates the intracellular sensors contained in the inflammasome complex of the keratinocytes, leading to the activation of caspase-1 and the processing and secretion of pro-inflammatory cytokines. Successive exposure results in the activation of immune cells residing in the tissues that induce and perpetuate the inflammatory response.16
Innate immune response cellsKeratinocytesThe keratinocytes are essential for the development of ACD, due to their abundance in the epidermis and to their role in the formation of the skin’s anatomical barrier function.3 Although the haptens can penetrate through the intact skin, the importance of an intact cutaneous barrier can limit the sensitivity to ACD, based on the observation that specific diseases that hinder the barrier function have an increased risk of sensitization through the application of topical medicines.3 The keratinocytes express TLRs and answer to hapten, activating the TLR4, such as the dinitrofluorobenzene (DNFB) and Ni2+.3,14
The keratinocytes secrete cytokines, such as IL-6, IL-10, IL-18, and TNF, in addition to IL-1β and IL-18.24 Of particular importance is the TNF, which, together with IL-1β and IL-18, is necessary for the maturation and migration of the DC induced by hapten, from the skin to the dLN.3,25
Dendritic CellsThe main function of the DCs is to recognize and process external antigens and migrate to the dLNs, where they present the antigens to naïve T cells.26 These are specialized cells and present subtypes, such as Dermal Dendritic Cells (dDCs), Plasmacytoid Dendritic Cells (pDCs), and Langerhans Cells (LCs), which are the only DCs in the epidermis.27 DCs remain in an immature state in the skin and, when the haptens penetrate the dermis and are captured by the dDCs,3 there is a release of pro-inflammatory cytokines, such as IL-1β and TNF-α, favoring the maturation and migration of dDCs to the dLNs, colonizing the paracortex regions.26,28,29
After the contact with the antigen in the dermis, the dDCs diminish their migration speed and adopt the morphology characterized by multiple dendrites. Although the LCs and dDCs have the capacity to present antigens, they show different kinetics.30 The presentation of the antigen is necessary in the sensitization and elicitation stages, which is important to evaluate the relevance of the subtypes of DCs in each stage. The depletion of DCs in the skin of sensitized mice increases the elicitation stage, which suggests a regulatory function in this stage performed by some subtype of DC.18
Among the populations of DCs, that which most produces type 1 IFN is pDC.16 pDCs produce all type 1 IFN subtypes, called IFN-α, IFN-β, IFN-l, IFN-w, and IFN-t.31,32 The type 1 IFNs resulting from pDCs activate the production of IL-12, IL-15, IL-18, and IL-23 by Myeloid Dendritic Cells (mDCs) and induce the monocytes to differentiate themselves into DCs. They also stimulate the NK cells and, together with IL-6, they lead the B cells to differentiate themselves into secreting plasmatic cells of antibodies.32-34 In addition, the pDCs increase the ability of the mDCs to carry out a cross presentation of exogenous antigens for the T CD8+ cells, promote their clonal explansion, and induce the differentiation of the Th1 cells.35
The langerina+ dDCs comprise a distinct subtype of DC derived from the precursors of bone marrow and represent a small percentage (~3%) of the total population of DCs in the dermis. Being in a dynamic state of flow, they are continually replaced by nine recruited cells.36 Another dermal population is the langerina- dDCs, which make up the majority of dDCs (~80%).3,36
With the evidence provided by langerina+ dDCs, the relevance of the epidermal LCs, such as APCs, have been questions in the cutaneous immune responses.8 The LCs were considered to be central cells in the presentation of antigens in the sensitization stage, as they possessed a potent ability to present antigens in vitro and because they were abundant in the epidermis. However, the langerina+ dDCs, and not the LCs, play an important role in sensitization, as they have sensitizing effects.18,26,37-39
NeutrophilsThe neutrophils can exercise an important antimicrobial and pro-inflammatory reaction through the production of ROS and are found in the inflammatory lesions of the skin of patients with ACD.40 A neutrophil infiltrate is detected in the skin just a few hours after sensitization and reaches a peak in approximately 24 hours.2 One deficiency of neutrophils in the sensitization can lead to a reduction in the induction and migration of DCs to the dLNs and to the deficiency in the induction of specific T cells toward the antigen.2 The neutrophils are involved in the stages of sensitization and elicitation of ACD, illustrating that they play a decisive role in the various stages, which was verified in a wide range of inflammatory models of skin allergy.2,41
Mast cellsMast cells are hematopoietically derived cells that reside for a long period of time in barrier tissues, such as the skin and the intestine. Mast cells express a high affinity to the IgE receptor (FcεRI), express TLRs 1-4, 6, 7, and 9, and respond to the various microbial products.42 In the reaction mediated by IgE, the immediate release of pre-formed granules containing histamine, proteases, proteoglycans, and TNF.3 The mast cells also secrete late pro-inflammatory mediators, such as IL-3, IL-4, IL-5, IL-6, IL-8, IL-9, IL-11, IL-13, TNF, and chemokines, such as CCL2, CCL3, and CCL4.3
During the sensitization stage, as they promote their own recruitment, the mast cells are required by the contact allergen, inducing local inflammation, activation, and migration of the DCs, and the generation of allergen-specific T cells.2 The contact allergens trigger the mast cells to secrete histamine, which itself acts in the endothelial cells and contributes to the recruitment and infiltration of neutrophils during the sensitization stage of ACD.18,43 The depletion or absence of mast cells before sensitization hinders the migration/maturation of CD8+ T cells,18,43 thus reducing the appearance of ACD.1,41
Natural Killer (NK) CellsNK cells are of lymphoid origin, but they do not reorganize the T-Cell Receptors (TCR) to recognize antigens.44 The NK cells accumulate in many inflammatory skin diseases, such as psoriasis, lichen planus, and ACD. In ACD, the NK cells represent only 10% of the lymphocyte infiltrate and are triggered by secreted cytokines in the location of contact due to the Th1 and Th17 cell infiltrate.44 There are two types of NK human cells, which are defined according to its functional properties, migratory capacity, and expression of the surface markers.45 The majority of circulating NK cells are CD56lowCD16+, express low levels of perforins, and have a strong cytotoxic capacity. By contrast, NK cells CD56highCD16- constitute 10-15% of the circulating NK lymphocytes; however, these are predominantly found in the secondary lymphoid organs, and store low levels of perforins, though they possess immunomodulatory functions and release high levels of cytokines.44,46
The subpopulation of NK cells CD56highCD16-CD62L- is specifically recruited in the cutaneous inflammations and directly aggravates the reactions of ACD, due to the release of IFN-γ and TNF-α and because they induce apoptosis of keratinocytes.41 The NK cells lead to a rapid cleavage of the adherence molecules, such as e-cadherins, and the loss of cohesion of keratinocytes and spongiosis.44,47
Since the NK cells originate from the lymph nodes, they return to the blood and migrate to the liver, where they remain as hepatic memory cells specific to the antigen, which have a long life and are in constant change with the blood to inspect the peripheral tissues.26,48 A new provocation of the skin with the same hapten leads to the recruitment and/or conservation of NK memory cells specific to the antigen and can quickly respond to the hapten for at least 3 to 4 months.48
γδ T cellsγδ T cells are T cells that express heterodimers of the γ and δ chains of TCR.3 These cells are present in tissues, such as the intestine and skin, without originating from lymphoid tissues; express limited or invariable TCRs; and show a phenotype that is similar to that of T cells, which allows them to react quickly to the provocation of antigens.16 In the skin, γδ T cells consist of a small proportion of the total T cells in the dermis (2-9%) and epidermis (1-10%).16 The localization in the epidermis favors the contact with the hapten during the sensitization stage of ACD3 and are capable of activating dDCs through the production of TNF and IFN-γ.16
The γδ T cells produce growth factors that are essential to healing wounds, such as the connective tissue growth factor, the fibroblast growth factor-9, and the keratinocyte growth factor.16,49 In addition, the γδ T and NK cells produce cytokines that are generally associated with the Th1, Th2, and Th17 cells.16
Innate Lymphoid Cells (ILCs)The ILC family includes not only the classic NK cytotoxic cells and the Lymphoid Tissue Inducer Cells (LTi), such as the non-cytotoxic ILC population.50,51 These are characterized by the classic morphology of lymphoid cells, but there is a lack of molecule expressions on the cell surface that identify other immune cell types.50 Members of the ILC family indicate that the NK cells and non-cytotoxic ILC helper are independent strains. The non-cytotoxic ILCs consist of three distinct groups: ILCs-1, ILCs-2, and ILCs-3, including LTi.52,53 In this sense, the non-cytotoxic ILC cells are capable of producing IFN-γ and TNF, and are involved in bacterial and parasitic intracellular immunity.54 The ILCs-2 produce the type 2 cytokines, such as IL-4, IL-5, IL-9, and IL-13, in response to the epitelial cytokines IL-25, IL-33, and Thymic Stromal Lymphopoetin (TSLP), as well as produce the epidermal growth factor receptor and promote the necessary inflammation for anthelmintic immunity, allergic inflammation, and tissue repair.50,55,56 Depending on the stimulus, the ILCs-3 produce IL-17A, IL-17F, IL-22, TNF, and Granulocyte-Macrophage Colony-Stimulating Factor (GM-CSF) and can promote antibacterial immunity, chronic inflammation, or tissue repair. The ILC-1, ILC-2, and ILC-3 subtypes exhibit a functional similarity to the T-helper cells in terms of the expression of cytokines and the potential effector function (Chart 3).51
Non-cytotoxic ILC groups
| ILC-1: | |
| • | Production of IFN-γ and TNF. |
| • | Bacterial and parasitic intracellular immunity. |
| ILC-2: | |
| • | Production of IL-4, IL-5, IL-9, and IL-13. |
| • | nthelmintic immunity, allergic inflammation, and tissue repair. |
| ILC-3: | |
| • | Production of IL-17A, IL-17F, IL-22, TNF, and GM-CSF (Granulocyte-Macrophage Colony-Stimulating Factor). |
| • | Antibacterial immunity, chronic inflammation, and tissue repair. |
The keratinocytes actively regulate the magnitude of the immune response through the release of chemotactic factors that influence the quality and magnitude of the lymphocyte infiltrate and synthesize chemokines, which are decisive in attracting T CD8+ and CD4+ lymphocytes, thus amplifying the reaction of ACD.44,57
In response to pro-inflammatory cytokines, such as IFN-γ and TNF-α, the keratinocytes produce the CXC chemokines. The CXCL9, CXCL10, and CXCL11 chemokines are chemokines specifically from Th1 cells and selectively attract neutrophils. CCL2 is capable of attracting monocytes, DCs, LCs, memory T cells, and NK cells.16 CCL20 attracts memory T cells and LCs in the direction of the skin.58 CCL27 is exclusively and constitutively expressed by keratinocytes and preferentially attracts resident cutaneous T lymphocytes that express the Cutaneous Lymphocyte-Associated Antigen (CLA) (Chart 4).16,59
Chemokines
| CXCL9, CXCL10, and CXCL11: | |
| • | Chemotactics of Th1 cells. |
| • | Attraction of neutrophils. |
| CCL2: | |
| • | Attraction of monocytes, DCs, LCs, memory T cells, and NK (Natural Killer) cells. |
| CCL20: | |
| • | Attraction of memory T cells and LCs toward the skin. |
| CCL27: | |
| • | Expressed by keratinocytes. |
| • | Attraction of resident cutaneous T lymphocytes that express CLA (Cutaneous Lymphocyte-Associated Antigen). |
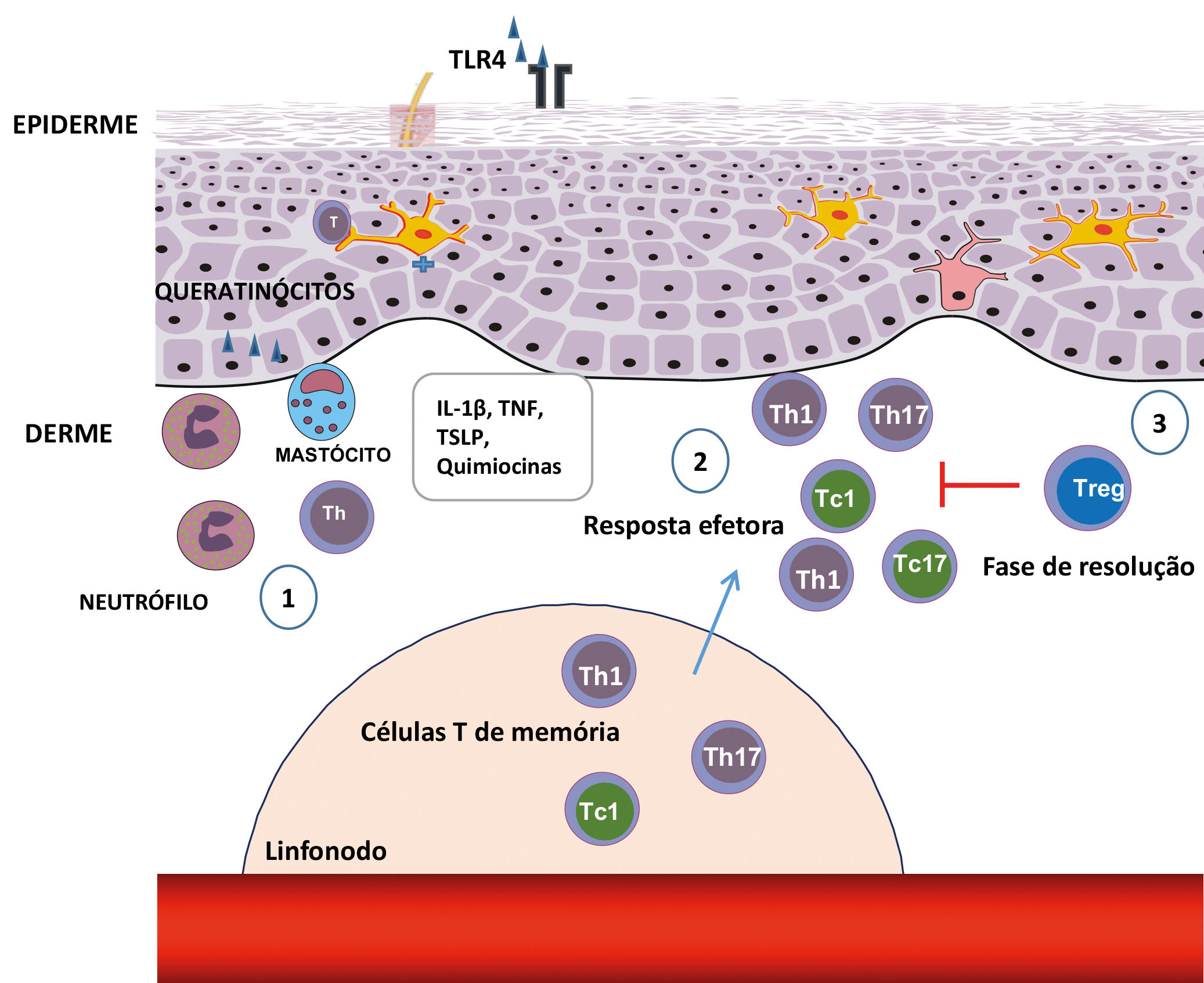
The mechanism of cellular infammatory infiltrate in the effector stage uses the inflammatory signals that are antigen-non-specific and antigen-specific.18
Antigen-non-specific inflammation - activation of keratinocytes, neutrophils, and mast cellsThe keratinocytes, neutrophils, and mast cells are essential factors to generate the antigen-non-specific inflammation in the elicitation or effector stage.18 First, the haptens stimulate the keratinocytes to produce pro-inflammatory cytokines, such as IL-1β and TNF-α. These cytokines activate the vascular endothelial cells to express adhesion molecules, such as ICAM-1 and P/E selectin, which guides the T cells in the blood to transmigrate to the tissues.23 The haptens also increase vascular permeability through histamine resulting from mast cells and trigger the keratinocytes to produce chemokines that aid the neutrophils to infiltrate into the skin, favoring the infiltration of the T CD8+ cells.41,43
When the concentration of the haptens is not raised enough to provoke the antigen-non-specific inflammation, the ACD reaction does not occur.18
Antigen-specific inflammation - T-cell activationFollowing the antigen-non-specific inflammation, the antigen-specific inflammation begins, mediated by T cells.18 When these infiltrate in the skin, they are activated by cutaneous APCs and produce IFN-γ and IL-17, which stimulate cells that reside in the skin, leading to the additional recruitment of T cells and intensifying inflammation.60 Each subtype of T cells (helper - Th and cytotoxic - Tc) trigger the cells in the skin differently and create their specific type of inflammation (Figure 2).18,61
Steps in the stages of elicitation and resolution of the contact dermatitis. (1) After reexposure to the haptens, antigen-specific T-cell infiltration occurs, favored by the production of chemokines and cytokines through keratinocytes, mast cells, and neutrophils. (2) The Th1, Th17, ou T CD8+ antigen-specific T cells, which secrete IFN-γ (Tc1) or IL-17 (Tc17), produce cytokines that induce the infiltration of additional inflammatory cells. (3) In the resolution stages, the Tregs infiltrate in the inflammatory sites and perform a suppressor function, contributing to the finalization of the inflammatory response
T CD8+ cells have pro-inflammatory effector functions, while the T CD4+ cells have pro-inflammatory and anti-inflammatory functions, which are dependent on the production of their cytokines or their subtypes.18
The effector cell is predominantly the T CD8+ lymphocyte, and the deletion of the Tc1 CD8+ cells has a greater suppressor effect than the deletion of Th1 CD4+ cells. Thus, the Th1 CD4+ and Tc1 CD8+ cells are effectors in ACD.62
The expansion of Ni2+-specific Tc1 CD8+ cells is decisive for the development of the contact allergy to Ni2+.6 However, the intensity of the inflammatory reaction is controlled mainly by the CD4+ effector T cells, which are predominant in the skin with ACD and release pro-inflammatory cytokines, in turn affecting the immune function of the resident cells.63
The role of Th1/Tc1, Th2/Tc2, and Th17/Tc17 cellsThe T CD4+ and T CD8+ cells can be divided into at least three relevant subtypes for the cutaneous imune response: Th1/Tc1, Th2/Tc2, and Th17/Tc17 cells. The Th1/Tc1 cells are characterized by the secretion of IFN-γ and IL-2; the Th2/Tc2 cells secrete IL-4, IL-5, and IL-1318,64; and the Th17/Tc17 cells cause the secretion of IL-6, TNF-a, GM-CSF, IL-17A, IL-17F, IL-21, IL-22, and IL-26 (Chart 5).64,65
The IFN-γ acts in the keratinocytes to produce Th1 cytokines, leading to the infiltration of inflammatory cells in the skin during the effector stage.18,66 IFN-γ and the cytokines produced by the T CD8+ (Tc1) and Th1 CD4+ cells promote ACD, while the type 2 cytokines, derived from Th2/Tc2 cells, negatively regulate them.18,62,66
The ACD caused by Ni2+ induces the Th17 cells, which are capable of stimulating pro-inflammatory cytokines, chemokines, and adhesion molecules produced by keratinocytes.67 IL-17 plays a stimulatory role in the sensitization and effector stages, and the IL-17 (Tc17) secretion cells are detected in the infiltrations of acute lesions and in all types of eczemas.7 The Ni2+-specific T CD4+ cells can secrete IL-17, and this shapes many pro-inflammatory functions of the keratinocytes, as well as acts in the recruitment of eosinophils for the eczematous cutaneous lesions.7,65,67
Resident T cells in the skin, such as immune sentinelsThe T cells that reside in the skin play an important role in immunological homeostasis and memory response.68 Memory T cells in the skin are strategically positioned as the first line of defense against the secondary provocation of the antigen and are important effector cells in the skin. Moreover, much like the T CD4+ cells, these are triggered by the skin’s DCs, resulting in the local proliferation of the antigen-specific CD8+ memory T cells.16,69
The pro-inflammatory cytokines and chemokines resulting from the T cells can promote a stimulus for the epithelial and mesenchymal cells, including keratinocytes and fibroblastos, thus amplifying the inflammatory reaction. In addition, the T cells in the skin can migrate to the epidermis and cross-attack an epithelial cell.16
Regulator ResponseRegulator T cellsThe Treg cells consist of a heterogeneous subfamily of T lymphocytes, which suppress the immune response through the release of anti-inflammatory cytokines or by deactivating the effector T cells through cell-cell contact, through the Cytotoxic T-Lymphocyte Antigen 4 (CTLA-4).26 These exist in all lymphoid tissues and include approximately 10% of all T cells in the normal skin, with a high proportion remaining in a resting state, especially in pilous follicles.70 Few can be found in the interfollicular dermis; the majority are located near the follicular epithelium.71
Tregs can be classified according to their origin: Natural Treg Cells (nTreg) of thymic origin and the Peripheral Treg Cells (pTreg). Both respond to antigenic recognition through their expansion, increase in suppressive capacity, and accumulation in the tissue where the antigen is located.72 They can suppress the activation, proliferation, and effector functions of a wide range of cells, such as CD4+, CD8+, NK, and APCs.73
Tregs exercise their regulatory function through the secretion of suppressor cytokines, especially IL-10, as well as by dependent factors of cell-cell contact, such as CD39/CD73 and granzyme/perforin.74,75 These act as suppressors of the activation of leucocytes, blocking their influz to the skin through the negative regulation of the selectin E/P expression in the endothelial blood cells.70,76 In addition to the direct suppressor action, Tregs indirectly suppress the proliferation of T cells by affecting the function of the APCs. These also negatively regulate the mast cells through the suppression of their degranulation and anaphylactic response.70,77,78
CD4+CD25+Foxp3+ Tregs are essential to the maintenance of self-tolerance perfomed by their regulator role, exercising the first line of control, while their depletion leads to an increase in T CD8+ cells, amplifying the ACD.4,70,79Forkhead box p3 (Foxp3) gene was identified as a transcriptional factor of the Tregs, and the memory cutaneous Tregs (mTregs) expressed high levels of Foxp3, when compared to mTregs in the peripheral blood.72,80,81 In addition to Treg Foxp3+, there are also other types of Tregs, such as T-regulator Tr1 cells and Th3 lymphocytes, which are induced in the periphery. It is possible that the Treg cells work in a system of cooperation.70
The Tr1 cells produce a large quantity of IL-10 and a moderate quantity of IL-5 and TGF-b, as well as restrict the differentiation and production of IL-12 through DCs, inhibiting the formation of effector and memory cells. These effects are mediated by the IL-10 and result in the suppression of the hapten-specific T CD4+ and CD8+ effector cells.26 The Th3 cells produce the antigen-specific TGF-b, exercising the suppressor effect.70
The peripheral T CD4+ lymphocytes of the individuals who are not allergic to nickel express a greater quantity of IL-10 and a lower quantity of IFN-γ than do allergic patients, that is, non-allergic individuals present a greater quantity of hapten-specific Tr1 cells in the blood and can also be found in the contact sensitivity skin lesions, in which the completion of the process is shaped.26
In the resting condition, a significant fraction (~20%) of T CD4+ cells derived from the skin in dLNs are Tregs.77 When the skin inflammation is induced after the application of hapten, one can observe a large number of Tregs derived from the skin in dLNs, with a strong capacity to inhibit the proliferation of hapten-specific T cells.82 These migrate more selectively in inflammatory conditions (effector stage) than in the stabilization/resting conditions18,83 and contain suppressor activities that are more potent than the Tregs found in dLNs.18,30,70,83 DCs, which express the antigen to the naïve T CD4+ cells form a stable cell-cell contact in the dLNs, and the Tregs interrupt this contact, mainly through their interaction with DCs.30
The Tregs capable of circulating toward the dLNs have a potential to remigrate to the skin, supporting the hypothesis that the skin can represent a lymphoid organ, in which the memory T-cell activities are modulated by the Tregs.18,30,83
The number of Tregs in the skin significantly increases during the cutaneous inflammatory process, suggesting that they contain a suppressive action in the inflamed location.18 Therefore, the cutaneous Tregs play an important role in terminating the ACD reaction and in the control of systemic immune responses.70
ConclusionThe development of ACD is unleashed by the cooperation of multiple factors, such as the environment in which the antigen appears, the type of antigen, and the genetic antecedentes. The cutaneous innate immunity acts in the activation of the adaptive immune response. Some contact allergens are detected by TLRs and NLR3 inflammasomes, acting in the induction and perpetuation of the inflammatory response. Many types of cells are involved in the innate immunity, such as: keratinocytes, DCs, neutrophils, mast cells, NK cells, gδ T, and ILCs, thus contributing to a sequence of immune response events, allowing for the early detection of contact allergens and the development of the local cutaneous inflammatory process. With the expansion of the effector T cells, there is the release of decisive pro-inflammatory cytokines for the development of the contact allergy. The Treg cells also play an essential role in maintaining tolerance and in the control of the inflammatory response. The number o Tregs in the skin increases during the cutaneous inflammatory process, suggesting that the Tregs play a suppressive role in the inflammed location, controlling the ACD reaction.
The initial immunological events of the development of ACD influence the maintenance or regulation of the contact reponse. Understanding how the contact allergens promote ACD is important not only to better understand the skin’s inflammatory process, but also in the prevention, diagnosis, and treatment strategies.
Financial support: None.
Conflict of interest: None.