Mycosis fungoides is the most common form of primary cutaneous lymphoma, with an indolent, slowly progressive course and 88% five-year survival rate. The diagnosis is challenging, especially in the early stages, and usually relies on a good clinical-histopathological correlation.
Objective:The aim was to establish the clinical and epidemiological profile of patients with early-stage mycosis fungoides.
Methods:This was a retrospective cross-sectional observational study with an exploratory analysis. Outcome variables were disease progression and mycosis fungoides-related death.
Results:One hundred and two patients were included. The majority were white males, with a mean age of 55.6 years. Mean time from onset of lesions to diagnosis was 51.08 months. The majority of patients were classified as IB stage according to TNMB. Mean follow-up time was 7.85 years. Disease progression was seen in 29.4% of the patients. Death related to the disease occurred in 7.9% of patients. Plaque lesions, involvement of more than 10% of the body surface, altered lactate dehydrogenase and beta-2-microglobulin, and stage IB were significantly associated with disease progression, and altered lactate dehydrogenase and beta-2-microglobulin also correlated with higher frequency of deaths.
Study limitations:Small sample and retrospective design.
Conclusions:The clinical and epidemiological profile of patients with early-stage mycosis fungoides in our sample corroborates reports in the literature. Diagnostic delay in our series is also consistent with previous findings, but the rate of disease progression, despite treatment, was higher than reported in the literature.
Primary cutaneous T-cell lymphomas (CTCLs) are a heterogeneous group of clonal T-cell lymphoproliferative disorders that affect the skin, without evidence of extracutaneous disease at the time of diagnosis. They account for 75-80% of all primary cutaneous lymphomas. Mycosis fungoides (MF) is the most prevalent CTCL, accounting for 50-65% of cases.1–5
Even so, MF is a rare disease. The incidence in the United States is approximately 0.3 to 1.02 new cases per 100,000 inhabitants/year.3,4 There are no data in Brazil on the incidence or prevalence of MF. The estimate for the year 2016 published by the Instituto Nacional de Câncer (INCA) for non-Hodgkin lymphomas (NHL) was 5,210 new cases in men and 5,030 in women, corresponding to an estimated risk of 5.27 new cases per 100,000 men and 4.88 per 100,000 women.5,6 Cutaneous lymphomas account for some 3.9% of all NHLs, and the majority of these are MF.1 However, the INCA statistics make no distinction in relation to MF.
MF typically affects individuals in their late fifties and predominates in males, with a sex ratio between 1.6:1 and 2:1. The majority of patients are Caucasian.1,5 The etiology is still uncertain, but it appears to result from chronic antigenic stimulation leading to T-lymphocyte clonal expansion and infiltration of the skin. However, the determinant or participating factors in this antigenic stimulation are still unknown.1,2
Diagnosis of MF is challenging, especially in the initial phases, and is usually based on clinical-pathological correlation. Immunophenotyping of the lymphoid infiltrate in the skin can provide additional information, and more recently, molecular methods have gained space in the research setting and are now available in clinical practice in large specialized centers.1,2,7
Pimpinelli et al. proposed a diagnostic algorithm (Chart 1) aimed at increasing the diagnostic accuracy, especially in early phases of the disease.7 The algorithm is a score based on clinical, histopathological, and immunohistochemical findings and T-cell receptor (TCR) gene rearrangement, where diagnosis of MF is defined as a total of at least four points. Use of this algorithm is supported by the International Society for Cutaneous Lymphoma (ISCL) and has been demonstrated in recently published review articles.1,2,8
Diagnostic algorithm for early stage mycosis fungoides
| Criteria | Score |
|---|---|
| Clinical criteria | |
| Basic | |
| - Patches or fine plaques with persistent and/or progressive evolution | 2 points: basic criterion + 2 additional |
| Additional | 1 point: basic criterion + 1 additional |
| - Non-photoexposed areas | |
| -Varying shape and size between lesions | |
| - Poikiloderma | |
| Histopathological criteria | |
| Basic | |
| -Superficial lymphoid infiltrate | 2 points: basic criterion + 2 additional |
| Additional | 1 point: basic criterion + 1 additional |
| -Epidermotropism without spongiosis | |
| - Lymphocytic atypia* | |
| Molecular criteria | |
| - Test for T-cell receptor clonal rearrangement | 1 point for clonality |
| Immunohistochemical criteria | |
| -<50% CD2+, CD3+, and/or CD5+ T-cells | 1 point per 1 criterion or more |
| -<10% CD7+ T-cells | |
| -Dermal-epidermal discordance in relation to CD2, CD3, CD5, and CD7 positivity** |
Total of four points defines diagnosis of early-stage mycosis fungoides; * Lymphocytic atypia defined as presence of large hyperchromatic, irregular or cerebriform nuclei.
Discordance refers basically to the absence of proper T-lymphocyte antigens in the cells contained in the epidermis. Adapted from Pimpinelli et al., 2005.7
The World Health Organization (WHO) and European Organization for Research and Treatment of Cancer (EORTC) classify MF in the group of indolent CTCLs, together with three of its variants: folliculotropic MF, pagetoid reticulosis, and granulomatous slack skin (GSS). It shows a chronic clinical course and progresses slowly, with 88% overall five-year survival.1,9
The staging proposed by the Mycosis Fungoides Cooperative Group (MFCG) and later revised by ISCL/EORTC is the main prognostic factor.10 Staging is based on the pattern of skin involvement, lymph node involvement, extension of the disease to other organs, and finally extension to peripheral blood, and is called TNMB (Charts 2 and 3). Patients in the initial stage are classified according to TNMB as stages IA (T1N0M0), IB (T2N0M0), or IIA (T1or2N1or2M0). Most patients are in the initial stage at the time of diagnosis.4,11
TNMB staging of MF-type CTCLs
| Cutaneous involvement | |
| T1 | Patches, papules, and/or plaques limited to 10% of body surface. T1a (patches)/T1b (patches and plaques). |
| T2 | Patches, papules, and/or plaques involving more than 10% of body surface. T2a (patches)/T2b (patches and plaques). |
| T3 | One or more tumors (greater than or equal to 1cm). |
| T4 | Confluence of erythema affecting at least 80% of body surface. |
| Lymph node involvement | |
| N0 | Absence of abnormalities in peripheral lymph nodes. |
| N1 | Presence of abnormal lymph node. Dutch histopathological grade I. N1a (clone-negative)/N1b (clone-positive). |
| N2 | Presence of abnormal lymph node. Dutch histopathological grade II. N2a (clone-negative)/N2b (clone-positive). |
| N3 | Presence of abnormal lymph node. Dutch histopathological grade III or IV. Clone-negative or positive. The Dutch histopathological classification of lymph node involvement is supported by the ISCL/EORTC. |
| Nx | Presence of abnormal lymph node, but without histological assessment/confirmation. |
| Visceral involvement | |
| M0 | Absence of visceral involvement. |
| M1 | Presence of visceral involvement, confirmed by histopathology. Must specify the organ involved. |
| Peripheral blood involvement | |
| B0 | Absence of peripheral blood involvement (less than or equal to 5% atypical lymphocytes or Sézary cells in peripheral blood). B0a (clone-negative)/B0b (clone-positive). |
| B1 | Presence of more than 5% atypical lymphocytes, but without fullfilling criterion for B2. B1a (clone-negative)/B1b (clone-positive). |
| B2 | Presence of 1,000 or more Sézary cells per mm3 in peripheral blood, clone-positive. |
Adapted from Olsen E et al., 2007.10
TNMB staging of MF-type CTCLs
| Stage | T | N | M | B |
|---|---|---|---|---|
| IA | 1 | 0 | 0 | 0 or 1 |
| IB | 2 | 0 | 0 | 0 or 1 |
| IIA | 1 or 2 | 1 or 2 | 0 | 0 or 1 |
| IIB | 3 | 0 or 2 | 0 | 0 or 1 |
| III | 4 | 0 or 2 | 0 | 0 or 1 |
| IIIA | 4 | 0 or 2 | 0 | 0 |
| IIIB | 4 | 0 or 2 | 0 | 1 |
| IVA1 | 1 or 4 | 0 or 2 | 0 | 2 |
| IVA2 | 1 or 4 | 3 | 0 | 0 or 2 |
| IVB | 1 or 4 | 0 or 3 | 1 | 0 or 2 |
Adapted from Olsen Eet al., 2007.10
This study aims to establish the clinical and epidemiological profile of patients with MF in the initial stages IA and IB, treated at the Photodermatology Sector of the Dermatology Service at Hospital Universitário Clementino Fraga Filho/Universidade Federal do Rio de Janeiro (HUCFF/UFRJ). The secondary objective was to identify the time from onset of the clinical lesions and diagnostic definition of MF, in addition to studying the disease progression rate (upstaging).
MethodsThis was an exploratory, observational, retrospective cross-sectional study. The study sample consisted of patients with a diagnosis of MF treated at the Photodermatology Sector of the HUCFF/UFRJ from January 2000 to December 2015.
Patients with MF were defined as those fullfilling the following three criteria:
Macules/patches (alterations of the skin involving only the color, with no change in the relief or consistency) or plaques (elevated flat lesions greater than 1cm in diameter) that are hypochromic, erythematous, or copper-red, with or without desquamation, of different sizes, involving mainly photoprotected areas;12
Chronic persistent or progressive evolution (at least six months);
Consistent histopathological examination (considering the clinical-pathological correlation). This situation includes the presence of lymphoid infiltrate along the dermal-epidermal junction, accompanied by the entry of lymphocytes in the epidermis, unaccompanied by spongiosis, or lymphocytic atypia (large hyperchromatic, irregular nuclei).
The analysis excluded patients with at least one of the following criteria:
Staging IIA or greater (disease with lymph node involvement, thus no longer limited to the skin);
Insufficient data on the patient’s medical record;
Positive serologies for human T-cell lymphotropic viruses (HTLV) 1/2;
Diagnosis of other associated lymphomas;
Follow-up time less than five years.
The dependent variables were:
Disease progression (upstaging), treated qualitatively, dichotomously, and nominally. Since the study included patients in stages IA or IB, disease progression was defined as patients that progressed to stage IIA or higher.
Death related to MF (either the lymphoma as the cause of death or deaths related to complications from the systemic therapies used). The variable MF-related death was also treated qualitatively, dichotomously, and nominally.
The independent variables were:
Gender, evaluated qualitatively, nominally, and dichotomously, considering the male or female sex of the study subject.
Skin color, described qualitatively and nominally as the self-declared color on the patient’s medical file, as white, brown, or black.
Age at diagnosis, follow-up time, and time of evolution of the disease, all analyzed quantitatively, continuously, with the first two in years and the latter in months.
Macules/patches (alterations of the skin involving only the color, with no change in the relief or consistency) or plaques (elevated flat lesions greater than 1cm in diameter),12 thus establishing qualitative and nominal evaluation. In the classification of this variable, there were more plaques than macules.
Topography of the lesions, differentiating between photoexposed and non-photoexposed areas, thus analyzed qualitatively, dichotomously, and nominally.
Number of lesions, assessed qualitatively, dichotomously, and nominally, differentiating between single and multiple lesions.
Percentage of affected body surface, treated qualitatively and ordinally, as less than 10%, 10 to 80%, and more than 80%.
Presence of lymphadenomegaly, hepatomegaly, and splenomegaly, assessed qualitatively, dichotomously, and nominally, as absence or presence in the patient.
Number of biopsies for the definitive diagnosis, treated as a discrete quantitative variable.
Peripheral eosinophilia and serum lactic dehydrogenase (LDH) and beta-2-microglobulin were also analyzed qualitatively, dichotomously, and nominally, and the results were thus classified as normal versus altered.
Imaging tests, when performed, were only differentiated as altered versus unaltered in relation to the lymphoma. Therefore, this was a qualitative, dichotomous, and nominal variable.
Possible comorbidities and medications of continuos use were analyzed descriptively. For medications of continuos use, we opted to record only the antihypertensive drugs for subsequent discussion of a recent article addressing the relationship between thiazide antihypertensives and MF.
The selected data were compiled on printed, digitized spreadsheets using Excel 2011 (Microsoft® Excel® for Mac 2011/Version: 14.2.0). Data were analyzed with SPSS, version 24.0. To analyze associations between the quantitative independent and dependent variables (disease progression and MF-related death), we used the Student t-test for independent samples or Mann-Whitney (non-parametric) test, and for qualitative independent variables we used the chi-square (X2) test or Fisher’s exact test.
Prevalence ratios and respective confidence intervals (95%CI) were calculated as the measures of association. Significance was set at 5%.
The study complies with Resolution 466/12 of the Brazilian National Health Council and is registered with the Brazil Platform. It was approved by the local Institutional Review Board (CEP-HUCFF/UFRJ: CAAE 59235916.9.0000.5257).
ResultsFrom a total of 135 patients identified in the registry of the Photodermatology Sector, 102 were included in the study following application of the inclusion and exclusion criteria.
Among the 33 excluded patient charts, 17 showed missing data or insufficient follow-up time, ten had a different diagnosis (parapsoriasis, lymphomatoid papulosis, or GSS), and six had positive serologies for HTLV 1/2.
Tables 1 to 6 show the results, subdivided in categories.
Clinical-epidemiological profile of the sample
| Gender | Male: 53.92%Female: 46.08% | 55/10247/102 |
| Skin color | White: 56.86%Brown: 30.40%Black: 12.74% | 58/10231/10213/102 |
| Age at diagnosis (years) | Mean: 55.16 (SD*: 15.97)Median: 57range: 10 to 83 | |
| Evolution (months) | Mean: 51.08 (SD: 63.00)Median: 33Range: 2 to 360 | |
| Primary lesion | Macules/patches: 45.09%Plaques: 54.91% | 46/10256/102 |
| Multiple lesions, various sizes | Yes: 99.02%No: 0.98% | 101/1021/102 |
| Scaling | Yes: 61.76%No: 38.24% | 63/10239/102 |
| Areainvolved | Photoexposed: 2.94%Non-photoexposed: 97.06% | 3/10299/102 |
| Surface area involved | <10%: 46.08%10 to 80%: 53.92%>80%: 0 | 47/10255/1020 |
| Lymphadenodomegaly | Yes: 6.80%No: 93.20% | 7/10295/102 |
| Hepatosplenomegaly | Yes: 0No: 100% | 0102/102 |
| Comorbidities: | Yes: 50.99%No: 49.01% | 52/10250/102 |
| Continuous medication | Yes: 35.30%No: 64.70% | 36/10266/102 |
| Number of biopsies for diagnosis | Mean: 2.52Median: 2Mode: 1Range: 1 to 7Distribution: 1: 28.43%/ 2: 26.47%/ 3: 23.52%/ 4: 11.76%/ 5: 5.88%/ 6: 2.94%/ 7: 0.98% |
All 102 patients entered the analysis for the variables age at diagnosis (years), evolution (months), and number of biopsies for diagnosis.
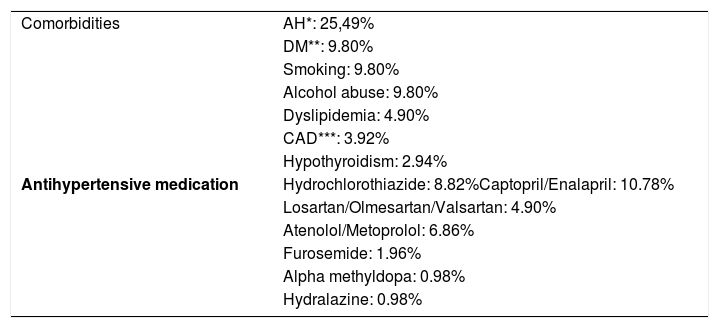
Comorbidities and continuous medication
| Comorbidities | AH*: 25,49% |
| DM**: 9.80% | |
| Smoking: 9.80% | |
| Alcohol abuse: 9.80% | |
| Dyslipidemia: 4.90% | |
| CAD***: 3.92% | |
| Hypothyroidism: 2.94% | |
| Antihypertensive medication | Hydrochlorothiazide: 8.82%Captopril/Enalapril: 10.78% |
| Losartan/Olmesartan/Valsartan: 4.90% | |
| Atenolol/Metoprolol: 6.86% | |
| Furosemide: 1.96% | |
| Alpha methyldopa: 0.98% | |
| Hydralazine: 0.98% |
Complementary tests
| Laboratory tests | ||
| - Peripheral eosinophilia | No: 81.37% | 83/102 |
| Yes: 18.62% | 19/102 | |
| - LDH | Normal: 64.04% | 57/89 |
| Elevated: 35.96% | 32/89 | |
| - Beta-2-microglobulin | Normal: 64.04% | 57/89 |
| Elevated: 35.96% | 32/89 | |
| - LDH+beta-2-microglobulina | Elevated: 20.22% | 18/89 |
| Imaging tests | ||
| - Chest x-ray | Normal: 100% | 94/102 |
| - Abdominal ultrasound | Normal: 100% | 92/102 |
| - Chest CT | Normal: 100% | 18/102 |
| - Abdominal and pelvic CT | Normal: 100% | 19/102 |
CT: computed tomography; LDH: lactic dehydrogenase
Patients’ follow-up and evolution
| Follow-up time (years) | Mean: 7.85 (SD*: 4.49)Median: 6Range: 2 to 24 years | |
| Disease progression | 29.4% (30/102) | Mean time to disease progression: 3.52 |
| MF-related death | 7.9% (8/102) |
Minimum follow-up time: five years. However, the analysis included patients that evolved to death during the period.
Prevalence ratios between independent and dependent variables
| Dependent variables | ||||||
|---|---|---|---|---|---|---|
| Progression | Death | |||||
| Independent variables | PR* | p | CI** | PR | p | CI |
| Primary lesion | Plaques | 1.3 | 0.013 | 1.061 – 1.745 | - | - |
| Surface area affected | 10 to 80% | 1.4 | 0.03 | 1.135 – 1.886 | 1.1 | 0.046 |
| LDH | Elevated | 2.1 | <0.001 | 1.400 – 3.164 | 1.2 | 0.010 |
| Beta-2-microglobulin | Elevated | 2.5 | <0.001 | 1.617 – 4.034 | 1.2 | 0.001 |
| LDH+beta-2-microglobulin | Elevated | 3.8 | <0.001 | 1.784 – 8.415 | 1.3 | 0.001 |
| Stage | IB | 1.3 | 0.008 | 1.091 – 1.780 | - | - |
There was a slight predominance of males (53.92%). Most patients in the sample were white (56.86%). Mean age at diagnosis was 55.16 years (standard deviation: 15.97), ranging from 10 to 83 years. Mean time from onset of lesions to diagnosis was 51.07 months (SD: 63.00), ranging from two to 360 months, as reported by the patients.
As for signs and symptoms, 54.91% of patients presented plaques as the predominant primary lesion. The absolute majority had multiple lesions of different sizes, except for one patient (0.98%) with a single lesion. Scaling was present in 61.76% of cases. Most patients had lesions predominantly on photoprotected areas, but in three patients (2.94%) the lesions predominated on photoexposed areas. In these, the lesions were more on the face, neck, and anterior thorax. In most patients (53.92%), between 10 and 80% of the skin was affected. We observed lymphadenomegaly, or slightly increased lymph nodes, with a fibroelastic consistency and regressing during follow-up, in 6.8% of the patients. No patient displayed hepatosplenomegaly.
Complementary tests and stagingAll patients had undergone a complete blood count at the time diagnosis, allowing to assess the differential leukocyte count and presence or absence of eosinophilia. However, 13 patients had no record of serum LDH or beta-2-microglobulin.
All patients in the sample underwent chest and abdominal imaging at the time of diagnosis.
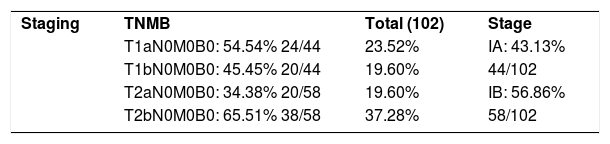
As for TNMB staging, 43.13% were classified as stage IA, and of these, 54.54% (23.52% of the total) were T1a, while 45.45% (19.60% of the total) were T1b. Thus, the majority of the patients (56.86%) were in stage IB at the time of diagnosis. Of these, 34.48% were T2a (19.60% of the total) and 65.51% were T2b (37.2% of the total).
Follow-up and evolutionOf the 30 patients that underwent disease progression, 33.33% advanced to IIA (developed N1); 16.66% to IIB (developed T3); and 40% to IIIA (T4). Of the three remaining patients, one progressed to stage IIIB (T4 + B1), on to IVA (T4 + B2), and one to stage IVB (M1).
Relationship between dependent and independent variables
Presence of plaque lesions, involvement of more than 10% of the body surface, altered LDH and beta-2-microglobulin, and stage IB were significantly associated with disease progression in the bivariate analysis. When related to death, only extent of the disease and altered LDH and beta-2-microglobulin remained significant. The other target variables were not statistically significant (Table 6).
DiscussionThis was a cross-sectional observational study aimed at characterizing the clinical-epidemiological profile of patients diagnosed with MF in the initial stage, seen at the Photodermatology Sector of HUCFF/UFRJ. In parallel, we assessed the mean time between onset of symptoms and diagnosis of the disease, besides the rate of disease progression (upstaging).
Corroborating the literature, most of the MF patients in this series were white males in their fifties. Two large cohorts published by Desai et al. and van Doorn et al., enrolling 393 and 309 patients, respectively, showed this same pattern for gender, mean age, and skin color.4,13
Mean time to MF diagnosis was 55.16 months, or some four years and a half between the onset of symptoms as reported by patients and a diagnostic definition. This time is obviously influenced by various factors, including the non-specificity of the initial symptoms, delay in securing specialized care, and difficulty in the clinical-pathological correlation. However, there is an evident need to shorten this time in order to benefit patients, since MF is a neoplastic lymphoproliferative disease. The mean number of biopsies to establish the diagnosis was 2.52, ranging from one to seven biopsies. This finding also objectively reflects the difficulty in diagnosing MF, especially in the initial stages.
A multicenter retrospective study with more than 13 years of follow-up showed a mean delay of four to six years between onset of symptoms and diagnosis of MF, a finding corroborated by other studies.13–15 In addition, diagnosis in the early stages is believed to depend on outpatient follow-up and multiple biopsies.8,14 Nikolaou et al. studied a population of 473 MF patients in which the mean time from onset of symptoms to diagnosis was 7.8 years. This same study showed a 3% increase in the risk of disease progression for each year in diagnostic delay after initial manifestation of the disease.16
Our clinical findings corroborate the clinical presentation of MF described in the literature: frequency of multiple lesions of different sizes (99.02%), with macules/patches (45.09%) and/or plaques (54.91%), mostly covered with desquamation (61.76%), affecting photoprotected areas (97.06%), with chronic, progressive evolution.1,2,3,5 This profile is consistent with that proposed by Pimpinelli et al. in their algorithm for the identification of initial-stage MF, such that only four patients in the sample (3.92%) would have scored only one point, while the vast majority of the rest would have met the maximum two points in the algorithm. This shows a good correlation between what we find in clinical practice and the algorithm’s proposal, emphasizing the importance of outpatient screening using these parameters.7
The majority of the patients presented some comorbidity, the most common of which was arterial hypertension. Among the hypertensive patients, 34.62% were in continuous use of hydrochlorothiazide. Continuous use of hydrochlorothiazide was found in 8.82% of the total sample, a higher rate than in the general population (5.8%).17 We chose to highlight this finding, even though it was not a specific objective of the study, since Jahan-Tigh et al., in 2013, found a temporal relationship between MF and hydrochlorothiazide, besides the fact that some patients reported improvement after suspending the medication, suggesting that hydrochlorothiazide could be a trigger for MF in a small group of patients.17 Further studies are necessary to verify the relationship between hydrochlorothiazide and MF.
Prior to the current study, the treatment protocol provided that all patients in the sample should be submitted to imaging tests at the time of diagnosis to complement the staging. The fact that no imaging test altered the staging defined previously by the clinical characteristics (i.e., all the imaging tests were normal in relation to extension of the lymphoma to other organs) might suggest a change in position in clinical practice. If there is no other clinical information to indicate complementary imaging, it could be waived at the initial workup.
Two large published cohorts found 78.3 and 71.5%, respectively, of patients in stage IA-IIA at the time of diagnosis.4,11 In the former, 43.2% (169/393) of patients were in stage IB, comprising the majority of the sample.4 In the latter, 31.16% were in stage IB, and in this series the largest share (40.20%) were classified as stage IA.4,11 The majority of patients in the current study were classified as IB (56.86%), with higher rates than described in the literature.
Mean follow-up time was 7.85 years. During follow-up, 29.40% of patients showed disease progression despite treatment. This rate was higher than in previous studies. Mean time from diagnosis to disease progression was 3.52 years (SD: 3.55) and the median was two years, ranging from six months (earliest upstaging) to 14 years (latest upstaging). Stratifying in relation to initial staging, 15.90% of IA patients and 39.65% IB of patients increased their staging. A cohort of 309 patients published by van Doorn et al. in 2000 showed five-year disease progression of 4% of patients in stage IA and 21% of patients in stage IB.13 More recently published case series have shown similar results to those of van Doorn et al.: 13% IA/27% IB upstaging4 and 25% encompassing stages IA and IB.15
Sixteen patients (15.68%) evolved to death during follow-up. Half of these (8 patients, thus 7.84% of the total) died due to disease progression of the lymphoma (4/8) or due directly to the chemotherapy (4/8). At five years, the percentage of deaths in the current study was lower than in the four cohorts cited previously.4,11,13,15
In addition to TNMB staging, knowledge of other factors can refine the prognostic workup of patients at increased risk of disease progression, which we find important, since even patients in the initial stage can evolve to advanced disease. Among the different target variables, the current study found that plaque lesions, involvement of more than 10% of body surface, altered LDH and beta-2-microglobulin, and stage IB were significantly related to disease progression, while altered LDH and beta-2-microglobulin also correlated with increased mortality. Studies have described the following factors with a clinical impact on prognosis: advanced age at diagnosis, male gender, plaque lesions, intense pruritis, lymphadenomegaly, peripheral eosinophilia, and elevated lactic dehydrogenase and beta-2-microglobulin.2,4,16,18-22
ConclusionWe conclude by emphasizing that the epidemiological profile of patients with MF in the initial stage, found in our sample, corroborates the findings in the literature. Our clinical findings also agree with the literature and overlap with the diagnostic algorithm for initial-stage MF proposed by Pimpinelli et al., so that we consider its use appropriate for defining cases. Despite advances in Dermatology, the mean time between onset of symptoms and diagnosis (although consistent with previous reports) still falls short of expectations in terms of early cancer diagnosis. Finally, the rate of disease progression was somewhat higher than reported by other authors. Mean time to diagnosis and the disease progression rate can be explained, at least in part, by our hospital’s characteristics, as a tertiary center with treatment determined by referral from primary and secondary services. The study’s limitations include the retrospective design and small sample.
AcknowledgmentsThe authors wish to thank Professor Nurimar Conceição Fernandes for her careful review of the manuscript.
Financial support: None.
Conflict of interest: None.